Veterinary physiology – Renal Tubular Reabsorption, Secretion and Excretion
Understand renal tubular reabsorption, secretion and excretion in veterinary physiology, including mechanisms, hormones and clinical relevance.
Renal tubular reabsorption, secretion and excretion are fundamental processes that determine the final composition of urine and maintain systemic homeostasis in animals. After glomerular filtration occurs in the renal corpuscle, the ultrafiltrate enters a complex tubular network where essential substances are recovered and unwanted solutes are eliminated. Understanding the physiology and pathophysiology of these processes is crucial for veterinary students, clinicians, and researchers, particularly when interpreting laboratory abnormalities or managing renal and systemic disorders.
In this article, we will explore the structural basis of tubular transport, the pathophysiology of solute handling, hormonal regulation, and the clinical implications of altered tubular function in veterinary medicine.
What Is Renal Tubular Reabsorption, Secretion and Excretion?
After filtration in the glomerulus, the ultrafiltrate formed in Bowman’s capsule closely resembles plasma in composition, except for large proteins. It contains:
Glucose
Amino acids
Electrolytes (Na⁺, K⁺, Cl⁻, HCO₃⁻)
Water
Small solutes
Without tubular reabsorption, the loss of these substances would be incompatible with life.
The nephron consists of:
Renal corpuscle (glomerulus + Bowman’s capsule)
Proximal convoluted tubule (PCT)
Loop of Henle
Distal convoluted tubule (DCT)
Connecting segment
Collecting duct
Each segment has distinct morphological and functional characteristics that determine its transport capacity.

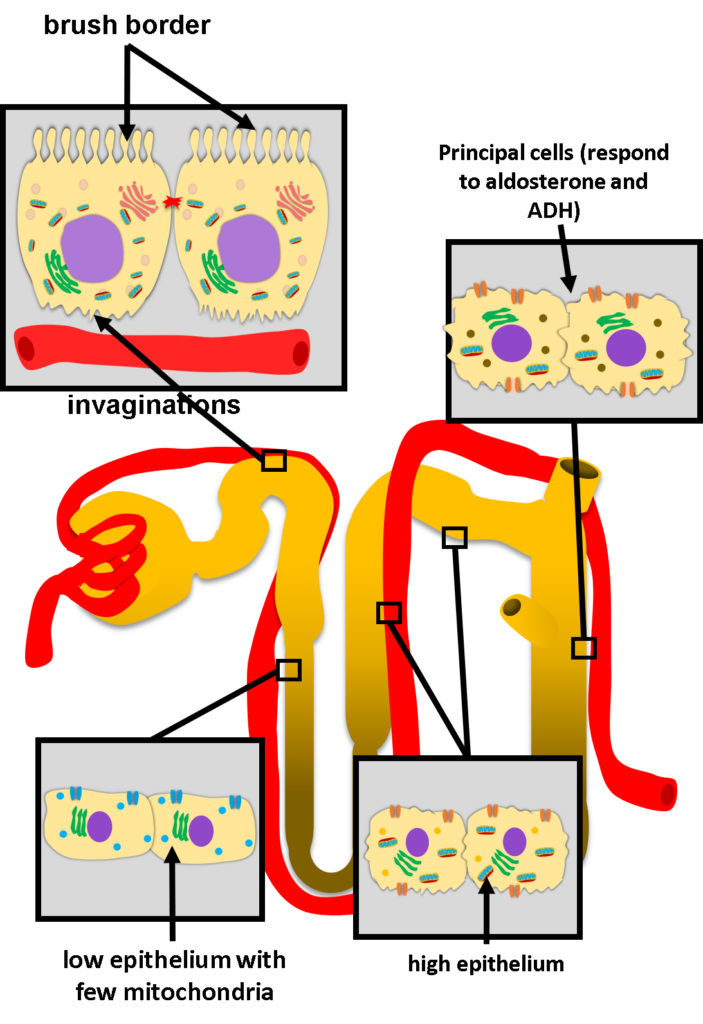
Structural Specialization of Tubular Segments
Proximal Convoluted Tubule (PCT)
The PCT is responsible for the majority of reabsorption. Its epithelial cells exhibit:
Prominent brush border (apical microvilli) to increase luminal surface area
Extensive basolateral membrane invaginations
High mitochondrial density for ATP production
These features support intense active transport and solute recovery.
Approximately:
65% of filtered water, sodium, and potassium are reabsorbed
100% of glucose and amino acids
50% of chloride
60–85% of bicarbonate
This segment performs bulk, relatively non-regulated reabsorption.
Loop of Henle
The thin descending limb has a simple, low epithelium with few mitochondria, reflecting passive transport characteristics.
The thick ascending limb contains taller cells rich in mitochondria and basolateral infoldings, enabling active sodium chloride transport. Importantly, the thick ascending limb and DCT are impermeable to water regardless of hydration status.
These structural differences are critical for medullary osmotic gradient formation and urine concentration mechanisms.
Distal Convoluted Tubule and Collecting Duct
The DCT continues active reabsorption of NaCl, calcium, and magnesium. It plays a key role in fine-tuning electrolyte balance.
The collecting duct contains:
Principal cells (respond to aldosterone and ADH)
Intercalated cells (acid–base regulation)
This segment adjusts urine composition according to the animal’s physiological state.
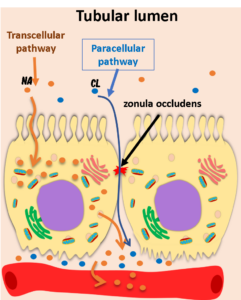
Mechanisms of Tubular Reabsorption
Reabsorption occurs through two primary pathways:
-
Transcellular pathway – across the apical membrane, through the cell, and out the basolateral membrane
-
Paracellular pathway – between cells through tight junctions (zonula occludens)
The Central Role of Sodium
Sodium is the driving force behind most tubular transport.
When filtered sodium reaches the proximal tubule, it diffuses into cells down its electrochemical gradient. On the basolateral side, the Na⁺/K⁺-ATPase pump actively transports sodium into the interstitium.
This creates:
-
A low intracellular sodium concentration
-
An electrochemical gradient
-
A slightly positive peritubular interstitial space
This gradient enables:
-
Cotransport of glucose and amino acids
-
Chloride reabsorption (often paracellular)
-
Osmotic water movement
Thus, sodium transport underlies the pathophysiology of fluid and electrolyte regulation.
Peritubular Capillary Dynamics
To understand how does tubular reabsorption occur efficiently, we must consider peritubular capillary forces.
During glomerular filtration:
-
Hydrostatic pressure drives fluid out of glomerular capillaries
-
Large proteins (e.g., albumin) remain in plasma
As filtration progresses:
-
Glomerular hydrostatic pressure decreases
-
Plasma oncotic pressure increases
When blood enters the efferent arteriole and peritubular capillaries, elevated oncotic pressure strongly favors fluid reabsorption into the capillaries.
This Starling force dynamic is essential for bulk proximal reabsorption.
Tubular Secretion: What Is It and How Does It Work?
Tubular secretion is often confused with excretion.
-
Secretion: transport of substances from peritubular capillaries into the tubular lumen
-
Excretion: final elimination of substances in urine
Many substances are protein-bound in plasma and therefore not filtered at the glomerulus. These include:
-
Creatinine (partially)
-
Prostaglandins
-
Catecholamines
-
Certain bile acids
-
Drugs (e.g., penicillins, diuretics)
In the proximal tubule, epithelial cells actively transport these substances from blood across the basolateral membrane and secrete them into the lumen.
This mechanism is clinically relevant in drug clearance and toxin elimination.
Segment-Specific Solute Handling
After the PCT, the epithelium transitions abruptly into the thin descending limb.
Loop of Henle
-
Descending limb: primarily water reabsorption
-
Thick ascending limb: active Na⁺, K⁺, Cl⁻ transport
-
Impermeable to water
Distal Convoluted Tubule
-
Active NaCl reabsorption
-
Calcium and magnesium handling
-
Water impermeable
Collecting Duct
-
Sodium reabsorption
-
Potassium secretion or reabsorption
-
Hormone-sensitive water transport
This distal regulation determines final urine concentration.
Hormonal Regulation of Tubular Function
Transport may be intrinsic or hormonally regulated.
Angiotensin II
Acts on:
-
Proximal tubule
-
Thick ascending limb
-
DCT
-
Collecting duct
It increases sodium reabsorption and is stimulated by decreased blood pressure.
Aldosterone
Acts on:
-
Connecting segment
-
Principal cells of collecting duct
Effects:
-
Increased sodium reabsorption
-
Increased potassium excretion
-
Indirect water retention
It is activated by hypovolemia and hypotension.
Antidiuretic Hormone (ADH)
ADH increases water permeability in the collecting duct by inserting aquaporins into the apical membrane.
Secreted in response to:
-
Dehydration
-
Hypovolemia
-
Hypotension
It concentrates urine and preserves body water.
Parathyroid Hormone (PTH)
Secreted in response to hypocalcemia.
Effects:
-
Decreases phosphate reabsorption in proximal tubule
-
Increases calcium reabsorption in thick ascending limb and DCT
-
Stimulates activation of vitamin D
Vitamin D
Enhances calcium reabsorption in DCT and connecting segment.
Calcitonin
Secreted during hypercalcemia.
Acts on:
-
Thick ascending limb
-
DCT
Decreases calcium and phosphate reabsorption.
Pathophysiology of Tubular Dysfunction
Disruption of renal tubular reabsorption, secretion and excretion can lead to:
-
Glycosuria without hyperglycemia (proximal tubular defect)
-
Metabolic acidosis (bicarbonate loss)
-
Hypokalemia or hyperkalemia
-
Isosthenuria
-
Drug accumulation
Tubular injury may result from ischemia, toxins, inflammation, or chronic kidney disease.
Understanding segment-specific transport allows clinicians to localize dysfunction.
Common Mistakes and Misconceptions
-
Confusing secretion with excretion
-
Assuming all reabsorption is hormonally regulated
-
Ignoring peritubular capillary oncotic pressure
-
Overlooking sodium’s central role in transport
-
Believing distal segments reabsorb large water volumes independent of ADH
Clarifying these concepts improves diagnostic reasoning.
Clinical Relevance Summary
Renal tubular reabsorption, secretion and excretion are central to:
-
Fluid balance
-
Electrolyte homeostasis
-
Acid–base regulation
-
Drug elimination
-
Blood pressure control
Segment-specific transport and hormonal regulation determine urine composition and systemic stability. Tubular dysfunction manifests through characteristic laboratory abnormalities, making physiological understanding essential for veterinary practice.
FAQ – Frequently Searched Questions
What is renal tubular reabsorption?
It is the process by which filtered substances are transported from the tubular lumen back into the bloodstream.
How does tubular secretion differ from excretion?
Secretion is movement from blood into the tubule; excretion is final elimination in urine.
What is the pathophysiology of proximal tubular dysfunction?
It involves impaired bulk reabsorption, leading to losses of glucose, amino acids, bicarbonate, and electrolytes.
What are the clinical signs of tubular injury in animals?
Polyuria, polydipsia, electrolyte imbalances, acid–base disturbances, and altered drug clearance.
How does ADH regulate water reabsorption?
ADH increases aquaporin insertion in collecting duct cells, enhancing water permeability and urine concentration.