Urine Concentration in Veterinary Physiology
Introduction
Urine concentration in veterinary physiology is one of the most critical mechanisms for maintaining body fluid balance in mammals. The kidneys continuously filter plasma, but most filtered water must be reabsorbed to preserve circulating volume and maintain homeostasis. The ability to produce hypertonic or hypotonic urine relative to plasma is essential for survival, especially in conditions of dehydration or excess water intake. Understanding how the nephron processes tubular fluid to generate concentrated or diluted urine is fundamental for interpreting urinalysis, assessing renal function, and applying pathophysiological reasoning in clinical practice.
When discussing urine concentration, we always compare urine osmolality to plasma. If urine is more concentrated than plasma, it is hypertonic. If less concentrated, it is hypotonic. If equal, it is isotonic. These physiological definitions are central to clinical nephrology and laboratory interpretation.
Nephron Types and Species Differences
Two main nephron populations exist:
Cortical nephrons
Juxtamedullary nephrons
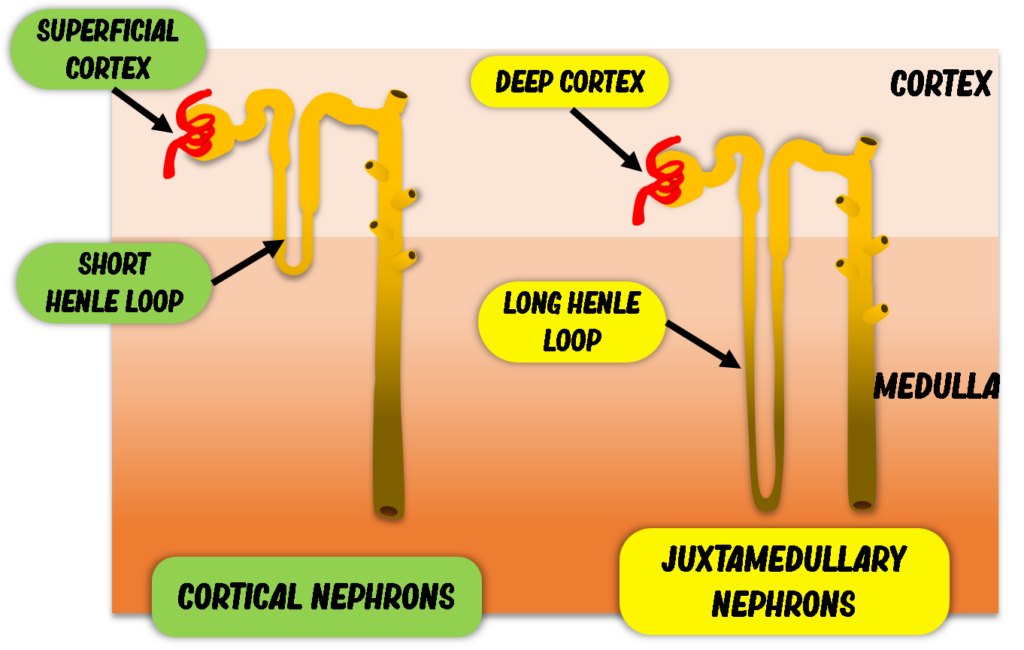
Juxtamedullary nephrons have long loops of Henle that extend deep into the medulla. This anatomical configuration is crucial for generating highly concentrated urine. Species variation plays a major role. Cats, for example, possess a very high proportion of juxtamedullary nephrons, which explains their remarkable ability to produce highly concentrated urine — an adaptation to low water intake.
The length and proportion of juxtamedullary nephrons directly influence maximal urine osmolality across species.
Anatomical Basis of the Countercurrent System
The structural organization of the loop of Henle and vasa recta underlies the kidney’s concentrating capacity.
Loop of Henle
The loop of Henle is U-shaped and consists of:
Thin descending limb
Thin or thick ascending limb
The descending limb is highly permeable to water but relatively impermeable to sodium chloride. In contrast, the thick ascending limb actively reabsorbs sodium and chloride via transporters such as NKCC2, and is impermeable to water.
Importantly, tubular fluid flows in opposite directions in the descending and ascending limbs. This opposing flow establishes the countercurrent multiplier mechanism, essential for creating a hypertonic medullary interstitium.
Vasa Recta
The vasa recta run parallel to the loop of Henle. Blood flow within these vessels occurs in the opposite direction to tubular fluid. These capillaries are permeable to water, sodium chloride, and urea, forming the countercurrent exchanger system.
This arrangement prevents washout of medullary hypertonicity while allowing reabsorbed water to return to systemic circulation.

Pathophysiology of Urine Concentration
What is the Medullary Osmotic Gradient?
For animals to generate concentrated urine, the renal medullary interstitium must be hypertonic. The deeper the medulla, the higher the osmolality. In many mammals:
Superficial medulla ≈ 300 mOsm/kg
Deep medulla ≈ up to 1200 mOsm/kg
This gradient is generated primarily by:
Active sodium chloride reabsorption in the thick ascending limb
Urea recycling in the inner medulla
Role of the Thick Ascending Limb
The thick ascending limb actively transports sodium and chloride into the interstitium but does not allow water to follow. This selective permeability is crucial. As solutes accumulate in the interstitium, the osmotic gradient increases progressively toward the inner medulla.
Because water cannot leave this segment, tubular fluid becomes progressively diluted as it ascends.
This segment is therefore known as a diluting segment.
Urea Recycling Mechanism
Urea plays a major role in maintaining medullary hypertonicity. In the inner medullary collecting duct, especially under antidiuretic hormone (ADH) influence, urea permeability increases. Urea diffuses into the interstitium, raising osmolality.
However, the thin descending limb is permeable to urea, allowing urea to re-enter tubular fluid. This creates a recycling loop that enhances water reabsorption efficiency.
Urea recycling is particularly important in mammals and contributes substantially to maximal urine concentration.
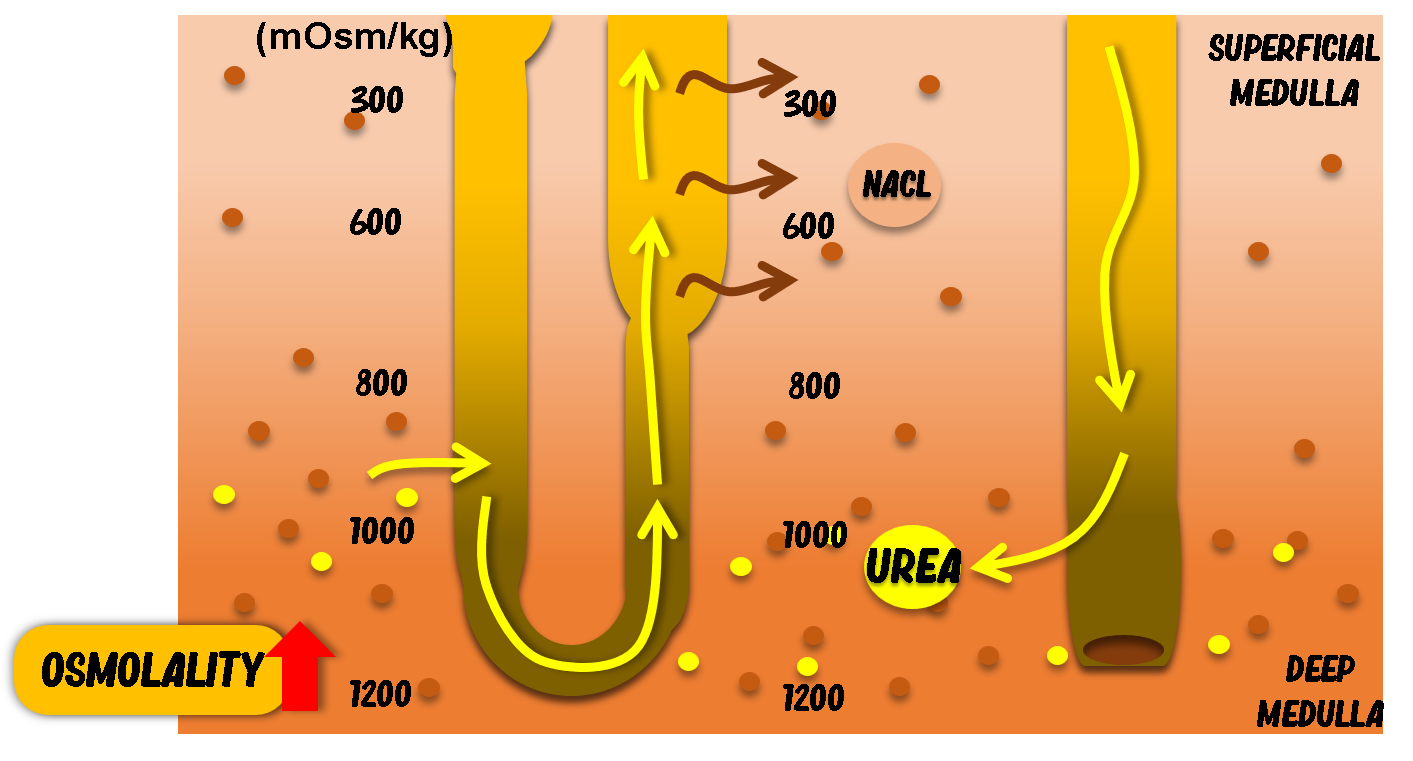
Segmental Handling of Water Along the Nephron
Proximal Convoluted Tubule (PCT)
More than 60% of filtered water is reabsorbed in the proximal tubule. This segment contains a brush border and abundant aquaporins, making it highly permeable to water.
Water reabsorption occurs isosmotically because solute reabsorption (especially sodium) drives osmotic water movement. Additionally, peritubular capillaries have high oncotic pressure due to retained plasma proteins, favoring water uptake.
At the end of the proximal tubule, tubular fluid remains isosmotic relative to plasma.
Descending Limb of Henle
As tubular fluid enters the descending limb and penetrates deeper into the medulla, water moves into the hypertonic interstitium by osmosis.
Because solutes remain in the tubular lumen while water leaves, fluid becomes progressively hyperosmotic relative to plasma.
This segment concentrates the tubular fluid.
Ascending Limb and Distal Convoluted Tubule
In the ascending limb and distal convoluted tubule, sodium chloride is reabsorbed but water cannot follow. This reduces tubular osmolality, making the fluid hypoosmotic by the time it reaches the collecting duct.
These are the principal diluting segments of the nephron.
Countercurrent Mechanism
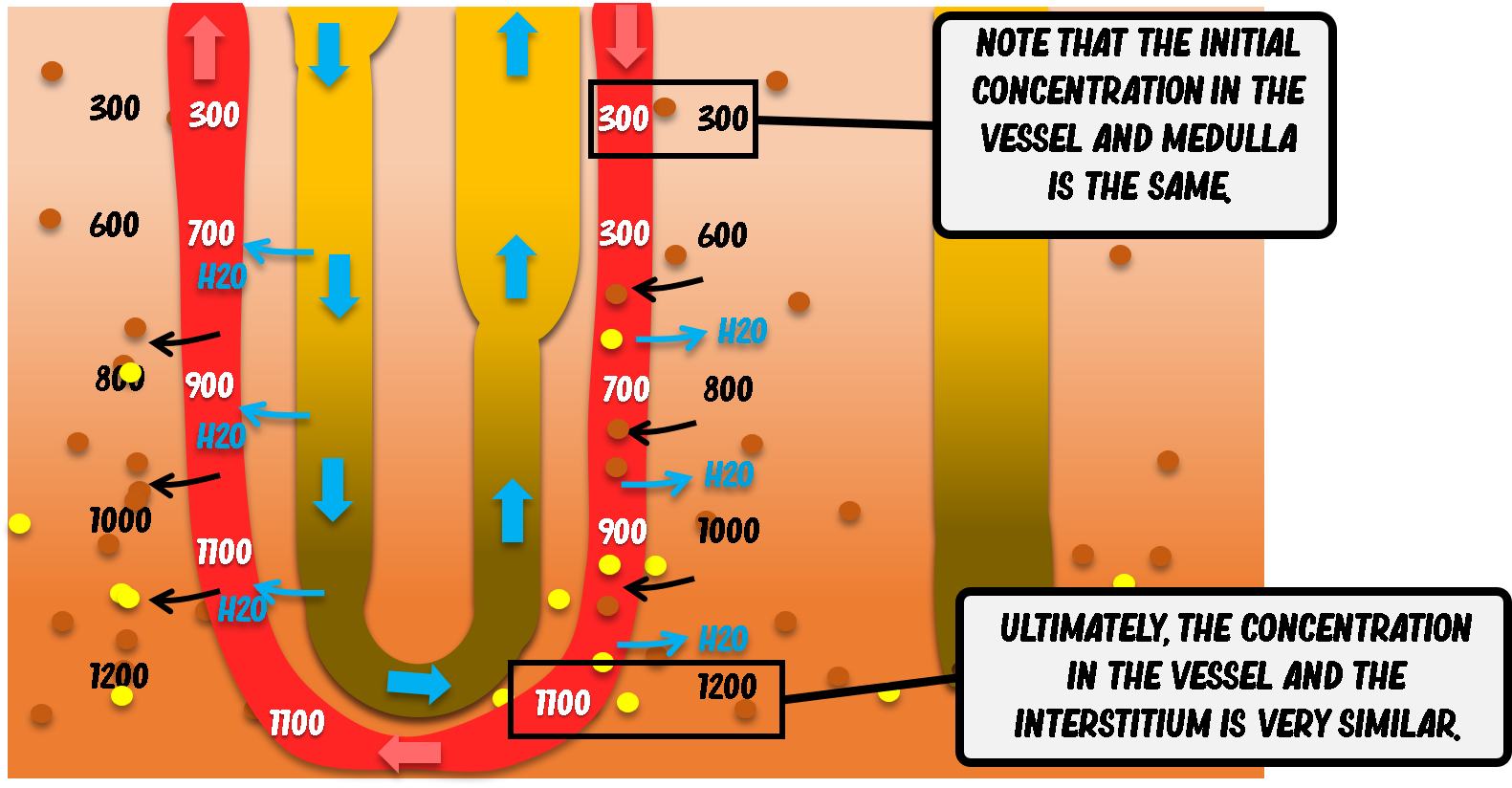
The vasa recta function as countercurrent exchangers, preserving the medullary osmotic gradient while removing the water reabsorbed from the loop of Henle and collecting duct. The image illustrates this dynamic process in detail.
At the point where blood enters the descending limb of the vasa recta, its osmolality is similar to that of the superficial medullary interstitium—approximately 300 mOsm/kg. At this level, there is minimal osmotic gradient between plasma and interstitium.
As the descending vasa recta penetrates deeper into the renal medulla, the surrounding interstitial environment becomes progressively more hypertonic (600, 800, 1000, up to about 1200 mOsm/kg). Because the vasa recta are highly permeable to water, sodium chloride, and urea, passive exchange occurs according to osmotic and concentration gradients.
As blood descends:
Water leaves the vessel by osmosis due to the increasing interstitial hypertonicity.
Sodium chloride and urea diffuse into the capillary, since their concentration is higher in the deep medullary interstitium than in the incoming plasma.
As a result, by the time blood reaches the deepest portion of the medulla, its osmolality closely matches that of the surrounding interstitium (approximately 1200 mOsm/kg), establishing local osmotic equilibrium.
When blood turns and begins ascending through the ascending limb of the vasa recta, the gradient progressively reverses. Now, the plasma is relatively hypertonic compared to the increasingly less concentrated interstitium of the outer medulla. Consequently:
Water moves back into the vessel by osmosis.
Sodium chloride and urea diffuse out of the blood and return to the interstitium.
By the time blood exits the medulla near the corticomedullary junction, its osmolality has returned close to its initial value.
This countercurrent exchange mechanism is essential because it prevents “washout” of the medullary gradient. If the vasa recta removed large amounts of solute without this controlled exchange, the hypertonicity of the medulla would dissipate. Instead, this system ensures that:
Reabsorbed water is returned to systemic circulation.
The medullary osmotic gradient is preserved.
The kidney maintains its ability to produce concentrated urine when required.
Importantly, the vasa recta do not generate the medullary gradient—that role belongs to the loop of Henle via the countercurrent multiplier mechanism—but they are indispensable for maintaining it.
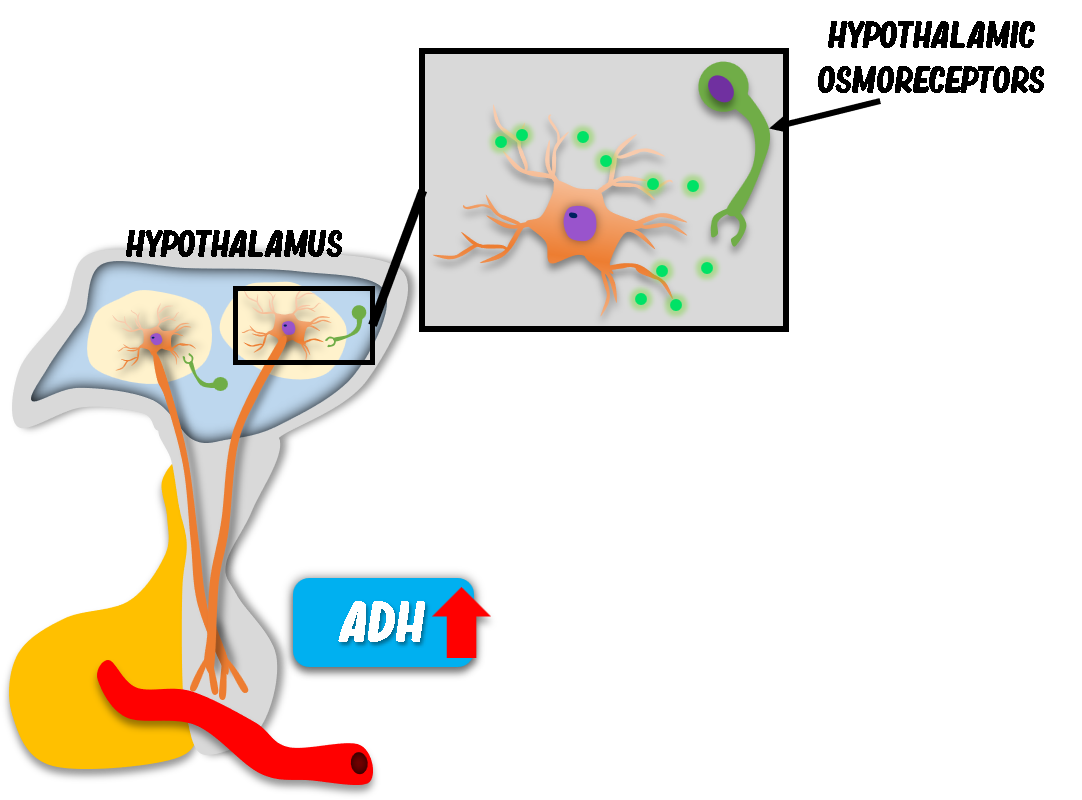
Role of ADH in Urine Concentration
How Does ADH Regulate Water Reabsorption?
The final determination of urine concentration occurs in the collecting duct.
In dehydration, hypothalamic osmoreceptors detect increased plasma osmolality. Additionally, angiotensin II stimulates ADH release. The posterior pituitary secretes antidiuretic hormone (ADH), also called vasopressin.
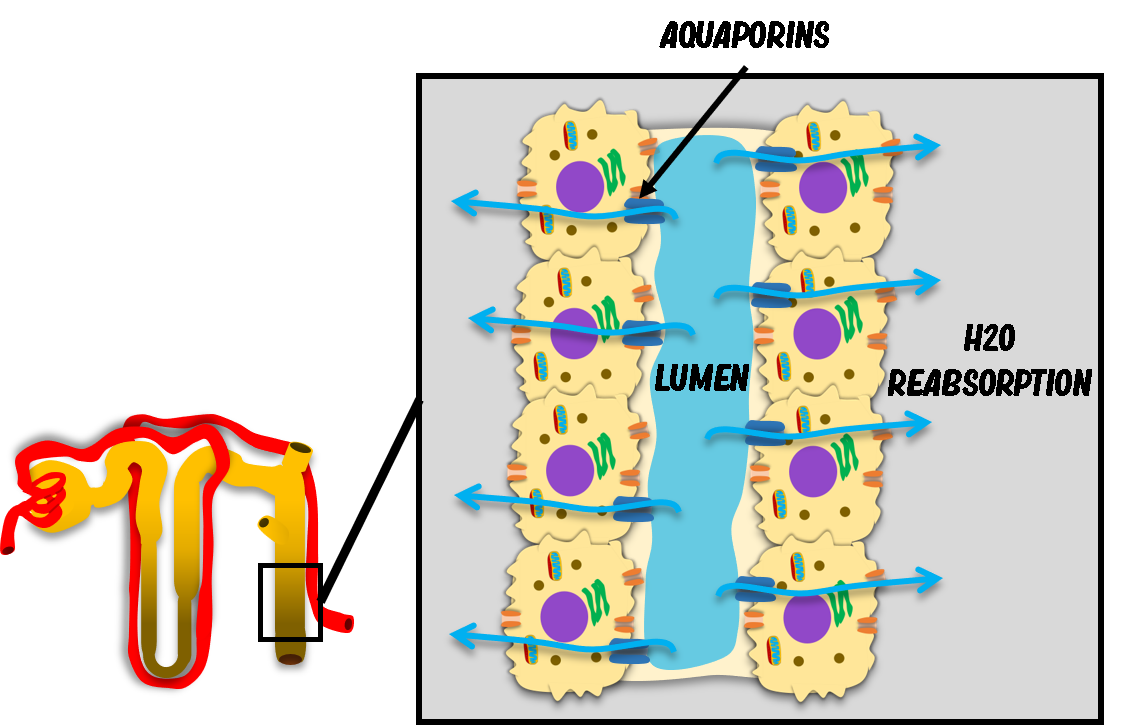
ADH acts primarily on principal cells of the collecting duct, inducing insertion of aquaporin-2 channels into the luminal membrane. This increases water permeability dramatically.
As water moves osmotically into the hypertonic medullary interstitium, urine becomes concentrated (hypertonic).
ADH also increases urea permeability in the inner medullary collecting duct, further enhancing medullary hypertonicity.
Absence of ADH
If ADH is not released, such as after excessive water intake, the collecting duct remains impermeable to water. The hypoosmotic tubular fluid formed in the ascending limb passes through unchanged, producing dilute (hypotonic) urine.
Clinical Correlation in Veterinary Medicine
Understanding urine concentration is fundamental for interpreting:
Urine specific gravity (USG)
Urine osmolality
Polyuria and polydipsia
Renal insufficiency
Endocrine disorders (e.g., diabetes insipidus, hyperadrenocorticism)
For example:
A dehydrated animal with low USG suggests renal concentrating failure.
A hypertonic urine in dehydration indicates intact ADH response and medullary gradient.
Diagnosis of Urine Concentrating Disorders
Conditions affecting urine concentration include:
Central diabetes insipidus (lack of ADH)
Nephrogenic diabetes insipidus (renal resistance to ADH)
Chronic kidney disease (loss of medullary gradient)
Medullary washout in prolonged polyuria
In chronic kidney disease, reduced nephron number and impaired countercurrent mechanism lead to isosthenuria (urine osmolality similar to plasma).
Practical Applications in Veterinary Routine
In daily clinical practice, evaluation of urine concentration should always be interpreted alongside hydration status.
Key considerations include:
Correlating USG with clinical dehydration
Assessing response to fluid therapy
Evaluating endocrine disorders
Monitoring progression of chronic kidney disease
Understanding the pathophysiology of urine concentration improves clinical reasoning and prevents misinterpretation of laboratory findings.
Common Mistakes or Misconceptions
Assuming concentrated urine always indicates normal renal function (it must match hydration status).
Ignoring species differences in concentrating ability.
Overlooking the role of urea in medullary hypertonicity.
Forgetting that tubular fluid is hypoosmotic when entering the collecting duct.
Confusing isosthenuria with hyposthenuria.
Clinical Relevance Summary
Urine concentration in veterinary physiology depends on:
A hypertonic medullary interstitium
Functional countercurrent multiplier and exchanger systems
Appropriate ADH secretion and renal response
Disruption of any component compromises water balance and may result in clinically significant polyuria, dehydration, or electrolyte imbalance.
A deep understanding of renal anatomy, physiology, and pathophysiology is essential for accurate interpretation of urinalysis and effective patient management.
FAQ – Searchable Questions
What is urine concentration in veterinary physiology?
Urine concentration refers to the kidney’s ability to produce urine that is hypertonic or hypotonic relative to plasma, depending on hydration status.
How does the loop of Henle concentrate urine?
The loop of Henle creates a medullary osmotic gradient via countercurrent multiplication, enabling water reabsorption in the presence of ADH.
What are the clinical signs of impaired urine concentration?
Polyuria, polydipsia, dehydration, and persistent low urine specific gravity are common findings.
How does ADH affect the collecting duct?
ADH increases water permeability by inserting aquaporin-2 channels and enhances urea reabsorption, promoting urine concentration.
What is the pathophysiology of isosthenuria?
Isosthenuria occurs when kidneys lose the ability to modify filtrate osmolality, typically due to chronic renal disease.